Introduction:
Chimeric antigen receptor (CAR) T-cell therapies have radically changed the treatment paradigm of patients (pts.) with relapsed/refractory (r/r) hematologic malignancies. Despite their remarkable efficacy, health care disparities can impact outcomes of pts. in the real-world setting. Living in a disadvantaged neighborhood has been associated with inferior outcomes after allogenic hematopoietic stem cell transplantation (HSCT), independent from individual-level socioeconomic (SE) factors. Our study aimed to identify the impact of neighborhood adversity on survival outcomes of recipients of CAR T-cell therapies, as this has not been yet described.
Methods:
We retrospectively identified consecutive adult pts. with r/r B-cell non-Hodgkin lymphoma (B-NHL) and multiple myeloma (MM) who received CAR T-cell therapies at our center from May 2018- January 2023. Neighborhood disadvantage was defined by Area Deprivation Index (ADI), a validated tool that allows for ranking of census block groups based on SE domains (income, education, employment, and housing quality) at a national level. The 2021 national ADI percentile ranks (1-100) were obtained for each pt. using the 9-digit zip code of place of residence, with a higher rank corresponding to higher level of disadvantage. We compared overall survival (OS) and progression free survival (PFS) between pts. with high vs. low ADI. We also examined survival outcomes in relation to estimated distance from place of residence to treatment center (DTC). Time dependent outcomes were calculated from date of CAR T infusion. Pts. with B-NHL and MM were analyzed separately. Baseline variables were compared between groups using chi-squared, t-test, and Wilcoxon rank sum tests as appropriate. Survival estimates were calculated with Kaplan-Meier method, and hazard ratios were generated using cox proportional hazards model.
Results:
CAR T therapy was administered in 124 and 45 pts. with B-NHL and MM, respectively. Median ages were 65 and 62 yrs., 68% and 51% were male, 94% and 84% were White, 47% and 20% had high-risk disease (based on IPI/MIPI for B-NHL, and FISH for MM), 30% and 69% had prior HSCT, and median number of prior therapies were 3 (range 1-6) and 6 (range 4-14), among pts. with B-NHL and MM, respectively. B-NHL subtypes included DLBCL (84%), MCL (6%), FL (5%), and PMBCL (5%), and CAR T products for B-NHL were axi-cel (52%), tisa-cel (24%), liso-cel (18%) and brexu-cel (6%). Ide-cel (82%) and cilta-cel (18%) were used in pts. with MM.
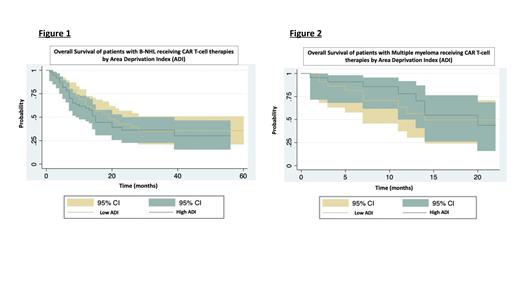
We treated pts. from 169 census block groups over 8 states, although 89% pts. were from Ohio. For the entire cohort, median ADI rank was 62.5 (range 1-100) and median DTC was 42.5 (range 1-4559) miles (m). Pts. were categorized (based on median) as having high (> 62.5) or low (≤ 62.5) ADI, and long (> 42.5m) or short (≤ 42.5m) DTC. Longer DTC was significantly associated with higher ADI ( p<0.001).
Baseline characteristics (age, sex, prior therapies, prior HSCT, high risk disease, CAR T product) were similar across groups with low vs. high ADI and short vs. long DTC. Median follow up time up was 12 (range 4-60) and 9 (range 4-22) months for living pts. with B-NHL and MM, respectively. For pts. with B-NHL, objective response rates (ORR) (85% vs. 80%; p=0.4), relapse rates (RR) (66% vs. 66%; p=0.9), median OS (19 vs. 14 months; HR of death: 1.2; 95% CI 0.7-1.8; p=0.5) ( Fig.1), and median PFS (10 vs. 5 months, HR of relapse: 1.1, 95% CI 0.7-1.7; p=0.6) were similar for pts. with low vs. high ADI. For pts. with MM, there were no differences in ORR (77% vs. 87%; p=0.3), RR (77% vs. 70%; p=0.5), median OS (14 vs. 18 months; HR of death: 0.8; 95% CI: 0.3-2; p=0.6) ( Fig.2), and median PFS (7 vs. 9 months, HR of relapse: 0.7, 95% CI 0.3-1.4; p=0.3) between those with low vs. high ADI. There were no differences in median OS of pts. with short vs. long DTC for NHL (19 vs. 14 months; HR: 1.2, 95% CI 0.7-1.9, p=0.4) or MM (14 vs. 20 months; HR: 0.9, 95% CI 0.3-2.5; p=0.9), or when DTC was analyzed as a continuous variable.
Conclusion:
In patients with r/r B-NHL and MM who received CAR T-cell therapies, response rates and survival outcomes were comparable regardless of neighborhood disadvantage level or distance to treatment center. Pts. living in more disadvantaged neighborhoods travelled longer distances for treatment. These findings prompt future investigation into referral patterns and access barriers to cellular immunotherapy, especially in disadvantaged neighborhoods.
Disclosures
Hamilton:NKARTA: Other: ad hoc advisory board; Angiocrine: Other: DSMB; Therakos: Honoraria; Equilium: Other: ad hoc advisory board; Kadmon/Sanofi: Other: advisory board; Incyte: Other: ad hoc consultancy; Rigel: Other: Ad hoc advisory board; CSL Behring: Other: Adjudication committee. Winter:Janssen: Consultancy; BeiGene: Consultancy; Seattle Genetics: Consultancy; AstraZeneca: Consultancy; ADC Therapeutics: Consultancy. Khouri:Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events; GPCR Therapeutics: Other: Payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events. Williams:Janssen: Consultancy; Bristol Meyers Squibb: Consultancy; Abbvie: Consultancy. Raza:Prothena: Honoraria; Pfizer: Honoraria; Kite Pharma: Honoraria; Autolus Therapeutics: Current holder of stock options in a privately-held company; ATARA Therapeutics: Current holder of stock options in a privately-held company. Jagadeesh:AstraZeneca: Research Funding; Daiichi Sankyo: Membership on an entity's Board of Directors or advisory committees; Affimed: Membership on an entity's Board of Directors or advisory committees; Trillium Pharmaceuticals: Research Funding; Seagen: Research Funding; Regeneron Pharmaceuticals: Research Funding; MEI Pharma: Research Funding; LOXO Pharmaceuticals: Research Funding; Debio Pharma: Research Funding; ATARA Biotherapeutics: Research Funding. Gerds:AbbVie, Bristol Myers Squibb, Constellation Pharmaceuticals, GlaxoSmithKline, Kartos, Novartis, PharmaEssentia, Sierra Oncology: Consultancy; Accurate Pharmaceuticals, Constellation Pharmaceuticals, CTI BioPharma, Imago BioSciences, Incyte Corporation, Kratos Pharmaceuticals: Research Funding. Brunstein:Allovir DSMB: Other: Consultant. Sauter:Kite/a Gilead Company, Celgene/BMS, Gamida Cell, Karyopharm Therapeutics, Ono Pharmaceuticals, MorphoSys, CSL Behring, Syncopation Life Sciences, CRISPR Therapeutics and GSK.: Consultancy; Juno Therapeutics, Celgene/BMS, Bristol-Myers Squibb, Precision Biosciences, Actinium Pharmaceuticals, Sanofi-Genzyme and NKARTA.: Research Funding. Hill:Incyte: Consultancy; Pharmacyclics: Consultancy, Other: Advisory board, Research Funding; Kite, a Gilead Company: Consultancy, Honoraria, Other: travel support, Research Funding; Genentech: Consultancy, Other: Advisory board, Research Funding; Bristol Myers Squibb: Consultancy; BeiGene: Consultancy; AstraZeneca: Consultancy; Gilead: Other: Advisory board; AbbVie: Consultancy, Other: Advisory board, Research Funding.